
The experiment that Rutherford designed is shown in Figure 22.2. However, using a radioactive source, a thin sheet of gold foil, and a phosphorescent screen, Rutherford would uncover something so great that he would later call it “the most incredible event that has ever happened to me in my life” When Ernest Rutherford began his gold foil experiment in 1909, it is unlikely that anyone would have expected that the plum pudding model would be challenged. More advanced students may be able to describe other models on their own. A chocolate chip ice cream model may work better to describe Thomson’s model to students. The plum pudding model is an outdated term many students will not understand. Though his understanding of our most elemental matter is rooted in the success of countless prior investigations, his surprising discovery about the interior of the atom is most fundamental in explaining so many well-known phenomena. While much could be written about any number of important scientific philosophers, this section will focus on the role played by Ernest Rutherford (1871–1937). 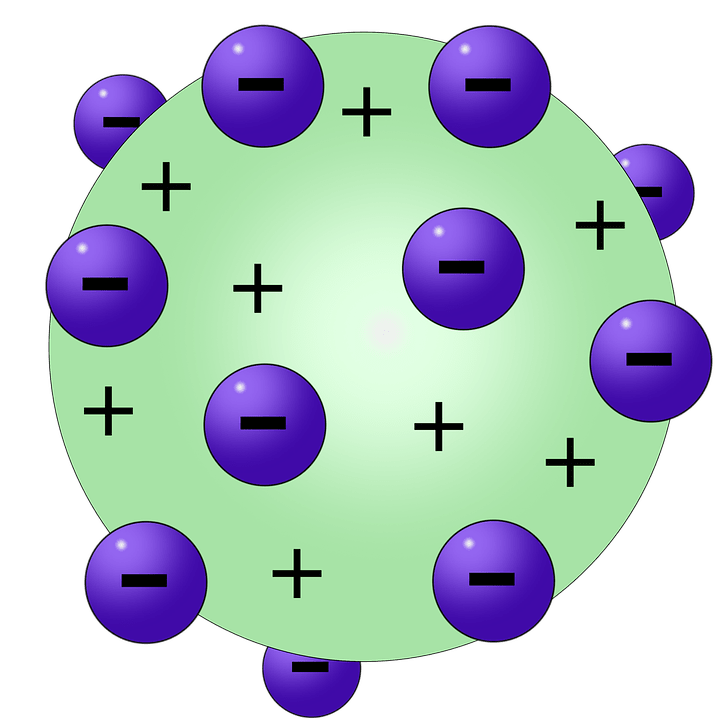
Since then, scientists such as John Dalton (1766–1844), Amadeo Avogadro (1776–1856), and Dmitri Mendeleev (1834–1907) helped to discover the properties of that fundamental structure of matter. 
The earliest known speculation on the atom dates back to the fifth century B.C., when Greek philosophers Leucippus and Democritus contemplated whether a substance could be divided without limit into ever smaller pieces. How do we know that atoms are really there if we cannot see them with our own eyes? While often taken for granted, our knowledge of the existence and structure of atoms is the result of centuries of contemplation and experimentation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
